Disposable Sterile Hypodermic Needle For Single Use
Product Features
| Intended use | The sterile hypodermic Needle for single use is intended for use with syringes and injection devices for general purpose fluid injection/aspiration. |
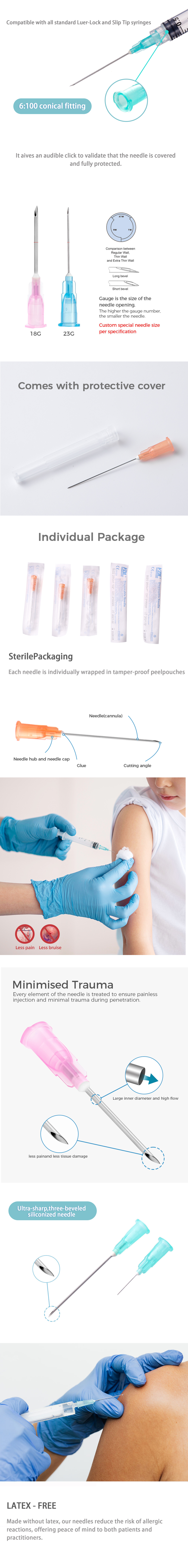
| Structure and compostion | Needle tube, Hub, Protective cap. |
| Main Material | SUS304, PP |
| Shelf life | 5 years |
| Certification and Quality Assurance | 510K Classification: Ⅱ
MDR(CE Class: IIa) |
Product Parameters
| Specification | Luer slip and Luer lock |
| Needle Size | 18G, 19G, 20G, 21G, 22G, 23G, 24G, 25G, 26G, 27G, 28G, 29G, 30G, 31G, 32G, 33G, 34G |
Product Introduction
Introducing our Sterile Disposable(hypodermic) needles, a reliable and essential device for medical professionals. These hypodermic needles are designed for ease of use, maximizing patient safety and ensuring each procedure is performed with precision and care.
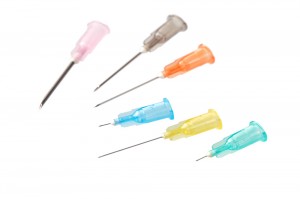
The hypodermic needles are available in a variety of sizes, including 18G, 19G, 20G, 21G, 22G, 23G, 24G, 25G, 26G, 27G, 28G, 29G, 30G,31G,32G,33G and 34G to meet diverse medical requirements. Featuring both Luer Slip and Luer Lock connections, they are compatible with a wide range of syringes and injection devices, making them suitable for general-purpose liquid injection and aspiration.
With a strong focus on quality and safety, these needles are made from non-toxic materials and properly sterilized to eliminate contaminants. Their single-use design ensures each needle is used only once, significantly reducing the risk of cross-infection and contamination.
Our products meet high industry standards, are FDA 510(k) cleared, and manufactured in compliance with ISO 13485 requirements. This demonstrates our commitment to strict quality control throughout the production process, ensuring every customer receives products of the highest quality.
In addition, our sterile hypodermic needles are classified as Class II under the FDA 510(k) regulation and comply with the EU MDR (CE Class IIa). This further confirms their reliability and safety in clinical use, giving healthcare practitioners full confidence.
In summary, KDL sterile hypodermic needles are essential medical devices due to their sterility, non-toxic materials, and full compliance with international industry standards. With our products, healthcare professionals can perform procedures confidently, relying on safe, reliable, and user-friendly devices that prioritize patient well-being.